|
5/15/2023 0 Comments Precipitate chemistry examples 
Adding a silver nitrate solution to aqueous solution of sodium chloride instantly produces white silver chloride and the solution produces sodium nitrate. And the reaction is called precipitation reaction (1) & (2). If one of the matters produced by the chemical reaction of two compounds in a solution is not dissolved in that solution and solidifies as a solid at the bottom of the vessel, it is called the precipitate. In precipitation, products are made by combining two separate solutions in aqueous solutions (1) & (2). These are burning, corrosion, cooking of food, eating, and much more. Some of these reactions we know and some we don’t know. In daily life, we see many types of chemical reactions. The following article is about what is precipitation in chemistry and its examples. 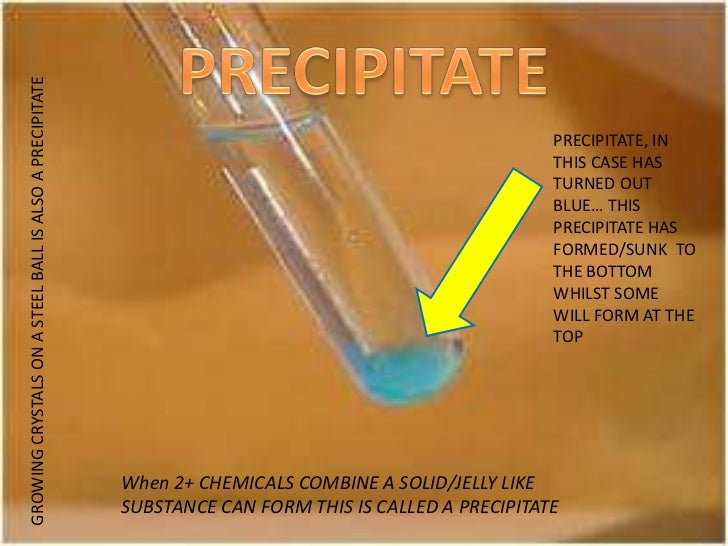
These reactions are divided into two parts a) Slow reaction b) Fast reaction. Some reactions are completed very quickly, while some reactions take a long time to complete. It takes some time for the reaction to be completed. Any chemical reaction is not complete as soon as it starts. The chemical reaction of matter does not happen by itself without a cause. 
Their properties are completely different from the properties of water. For example, electrolysis of water mixed with a small amount of acid dissolves the water and produces hydrogen and oxygen. A precipitation reaction is a part of double- displacement reactionĪ chemical reaction is a reaction that changes the molecular structure of matter resulting in the formation of new matter with one or more different properties. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
